RNA has emerged as both a powerful research tool and a promising therapeutic agent. From personalized cancer vaccines to gene editing and protein replacement therapies, the potential of RNA-based science continues to expand rapidly. As with any biological molecule, however, the quality of your RNA matters, and understanding the impurities that can arise during synthesis is an important part of getting the most out of your work.
What Are RNA 'Length' Impurities?
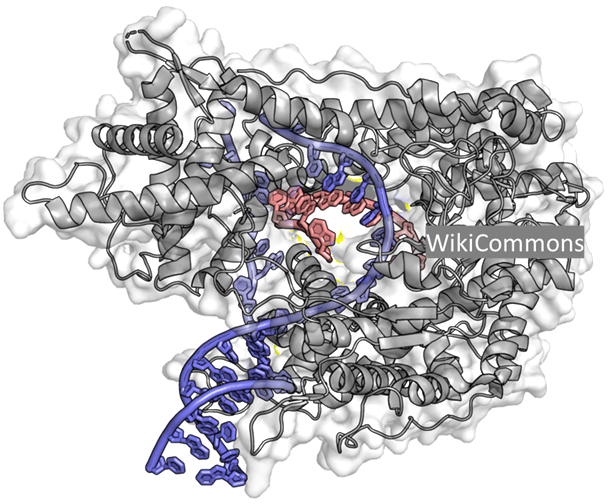
When RNA is produced using in vitro transcription (IVT), the RNA polymerases generally do not generate a single product. While the majority of transcripts are full-length, the process also produces a range of unwanted species, what we collectively call as 'length' impurities. These impurities are often present as a small percentage of the total RNA generated, and may not even be detected by standard analytical methods like chromatography. Yet in certain applications, even small amounts of these impurities can be consequential. Because RNA molecules are inherently “sticky”, they readily interact with other nucleic acid species already present or being generated during IVT, which contributes to the persistence of these impurities.
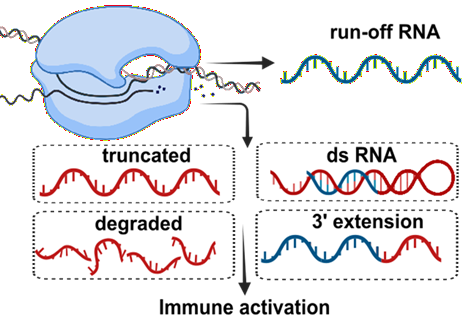
As shown in Figure 1, they include:
Abortive or truncated transcripts short, incomplete RNA fragments produced when the polymerase detaches prematurely during transcription. Degraded RNA fragmented transcripts resulting from RNA instability or nuclease activity during or after synthesis. Double-stranded RNA (dsRNA) a byproduct of the transcription process in which the RNA polymerase uses the newly synthesized RNA as a template or abortive transcripts already present to produce a long, usually 10s or 100s of nucleotides, complementary RNA strand. 3' extensions Short additional nucleotides added beyond the intended transcript end, altering the RNA's functional properties. RNA:DNA hybrids forms when the RNA transcript remains associated with the DNA template. Together, these constitute what are known as product-related impurities, all arising because of RNA's inherently sticky nature. Importantly, the problem scales with length. As RNA length increases, so does the percentage of these length impurities, making purity an increasingly relevant consideration for longer therapeutic mRNAs.
Why It May Matter for Your Research
The relevance of RNA impurities depends very much on your application. Some experiments may tolerate a degree of impurity, particularly where qualitative data, a simple yes or no for expression for example, is the goal. However, for applications requiring quantitative, highly reproducible, or high-dose applications, impurity levels can become a meaningful variable (Figure 2). In gene editing applications using mRNA for example, dsRNA impurities may activate innate immune pathways, triggering a cellular stress response that may reduce editing efficiency and may generate off-target effects. In protein expression studies, abortive and truncated transcripts may compete with your target mRNA for ribosomes, potentially reducing yield and introducing variability into your results. As shown in Figure 3, this effect becomes especially pronounced at higher mRNA concentrations, where impurities start to significantly affect protein expression levels. In diagnostics, impurities may interfere with assay sensitivity and specificity, introducing noise that affects the reliability of your results. In therapeutic development, purity requirements are at their most stringent. Immunogenic impurities such as dsRNA may pose safety considerations, and for high-dose mRNA therapeutics, even trace levels of dsRNA can trigger significant immune activation, potentially affecting a clinical program. These are important factors to account for early in development.
The Purification Challenge
Achieving high RNA purity is not straightforward. Producing pure RNA may require repeated and costly process development cycles, and even after significant investment in optimization, the resulting product may still not meet purity requirements. This iterative process may consume time and resources with no guaranteed outcome. HPLC has been shown to reduce or even eliminate length impurities from RNA preparations and can be considered as a gold standard for RNA purity. However, HPLC is currently used primarily as an analytical tool or for small-scale preparations in research settings, not as a routine, scalable method, leaving a gap between what can be measured and what can be reliably achieved in production. Cisterna's proprietary SPECC (SPEcific Capture and Cleavage) technology selectively and consistently produces full-length target RNAs, approaching HPLC-level purity without the associated bottlenecks. This level of purity means you can trust your RNA and build on your results with confidence as you move through R&D and beyond.
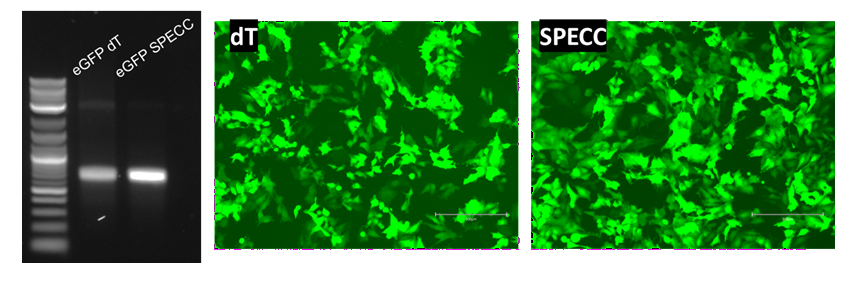
The functional impact of purity is further demonstrated in Figure 3, using mRNA encoding firefly luciferase as a model system.
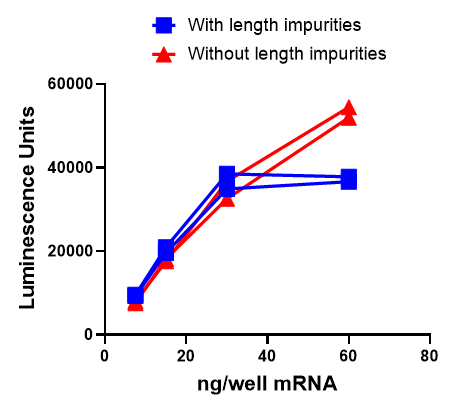
As the data show, at lower mRNA concentrations the difference is modest. But as concentration increases, the inhibitory effect of length impurities becomes clear: protein expressions plateau and suppressed in length-impurity-containing samples, while length-impurity-free mRNA continues to drive higher protein output. For any application where dose matters, this distinction can be significant.
Purity in Context
RNA purity is not a one-size-fits-all requirement. For qualitative assays or exploratory experiments, standard synthesis methods may be entirely sufficient. But for applications demanding the highest expression, reproducibility, or therapeutic safety, particularly in high-dose settings, purity may be paramount.
At Cisterna, we believe researchers should have access to RNA they can trust, without the barriers typically associated with high-purity production.
Interested in learning more about how Cisterna's SPECC technology works? Explore our technology or get in touch, we'd be happy to discuss your specific application.